A stem cell route to the roots of MND
This is a guest post by Nina Rzechorzek. The views, opinions and positions expressed are those of the writer, and may not necessarily represent those of EuroStemCell or other site contributors.
 It was a typical morning – trying to juggle experiments, trying not to make mistakes, trying hard to get results….sometimes life can be very ‘trying’ indeed… but then I’m not affected by motor neurone disease (MND) - and what a privilege it is for me to be able to rush around, to go to work and, hopefully one day, discover something that can make a difference. I am reminded of this as I stumble out of the morning into a less ordinary afternoon - stepping away from the bench and into the world of my boss, Prof Siddharthan Chandran.
It was a typical morning – trying to juggle experiments, trying not to make mistakes, trying hard to get results….sometimes life can be very ‘trying’ indeed… but then I’m not affected by motor neurone disease (MND) - and what a privilege it is for me to be able to rush around, to go to work and, hopefully one day, discover something that can make a difference. I am reminded of this as I stumble out of the morning into a less ordinary afternoon - stepping away from the bench and into the world of my boss, Prof Siddharthan Chandran.
Prof Chandran and his colleagues at The University of Edinburgh have created a hive of activity around research into all sorts of neurodegenerative diseases, including MND. At the heart of this hub sits The Anne Rowling Regenerative Neurology Clinic – a seamless interface between patients, clinicians and scientists. It is here that we listen and talk, we try to understand the challenges faced by patients and explain those that we see in our work. This is integral to translating stem cell research into new therapies. We need to make sure that the questions we ask are relevant to the disorders we hope to treat. Equally it is an opportunity to update patients on where the research has got to, and what it means for them.
So how can stem cells help?
Stem cells can be useful to MND research in 3 principal applications:
- studying diseases processes by ‘modelling’ them in a dish,
- drug screening and
- cell replacement therapy.
I will give a brief overview of each, with examples of how this is being put into practice.
‘We are the model’1
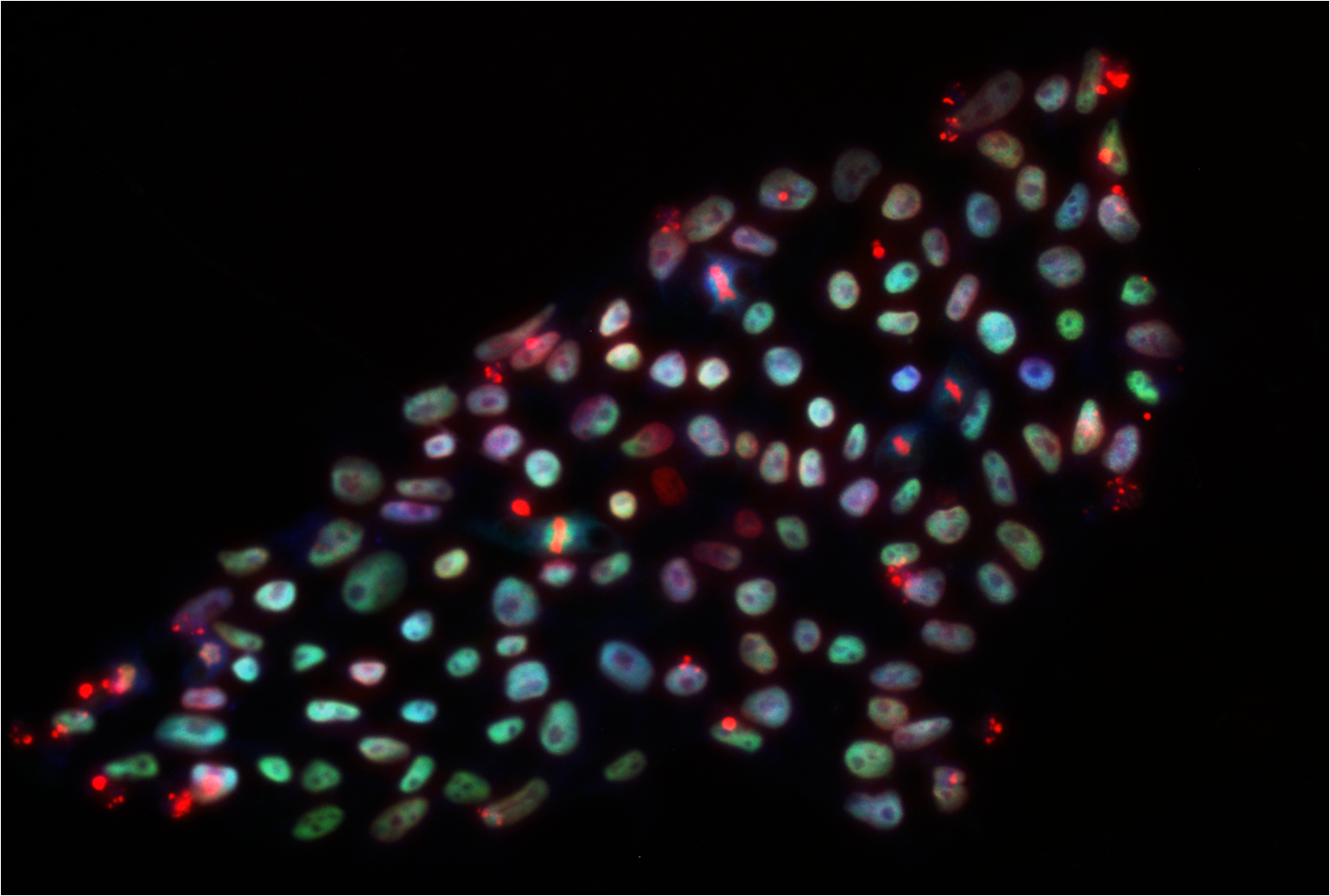 Several drugs that have shown promise in laboratory animal models of MND have made it through to human clinical trials (more than 30 in the last 15 years2). And yet, no effective treatments have emerged. Part of the problem is that MND does not result from one single cause. It involves many different types of cell, including the motor nerves that instruct our muscles to contract, the ‘nerve support cells’ or ‘glia’ - and even the muscle cells themselves. We are only beginning to understand how these cells interact with each other, both in health and throughout the course of MND. It remains unclear which, if any, of these cells are driving the disease process, or which of them might be effectively targeted for treatment. The Nobel-Prize-winning finding of Shinya Yamanaka and his colleagues in 20063 - that skin cells could be ‘reprogrammed’ into stem cells by manipulating just 4 genes - has revolutionized the way we approach disease modelling in the lab. These reprogrammed cells, or ‘induced’ pluripotent stem cells (iPS), can be used to generate any type of cell found in the body. For us, this means we can use a skin sample from an MND patient to grow motor nerves, glia and muscle cells – separately and together – in a dish. We can then use these cells to model aspects of MND-associated biology and what might go wrong with these cells in patients. We can study how the cells behave in isolation and how they interact with each other. For example, recent work showed that glial cells generated in the lab from a patient with an inherited form of MND were themselves affected by the inherited gene mutation, irrespective of any effect on motor nerves4. Many researchers are now using MND patient-derived stem cells like this to gain insight into disease processes. However, there are still teething issues to overcome. Keeping specialised human cells alive in a dish long enough for them to mature and for us to perform meaningful experiments can be very challenging5. Nonetheless, useful results are already emerging from iPS cell research. These include the discovery of a common genetic abnormality that links some cases of sporadic (non-inherited) MND with many cases of familial (inherited) MND6. This finding has shifted the focus of current laboratory work and already, iPS technology is being used to ask how this genetic abnormality might lead to the death of motor nerves7.
Several drugs that have shown promise in laboratory animal models of MND have made it through to human clinical trials (more than 30 in the last 15 years2). And yet, no effective treatments have emerged. Part of the problem is that MND does not result from one single cause. It involves many different types of cell, including the motor nerves that instruct our muscles to contract, the ‘nerve support cells’ or ‘glia’ - and even the muscle cells themselves. We are only beginning to understand how these cells interact with each other, both in health and throughout the course of MND. It remains unclear which, if any, of these cells are driving the disease process, or which of them might be effectively targeted for treatment. The Nobel-Prize-winning finding of Shinya Yamanaka and his colleagues in 20063 - that skin cells could be ‘reprogrammed’ into stem cells by manipulating just 4 genes - has revolutionized the way we approach disease modelling in the lab. These reprogrammed cells, or ‘induced’ pluripotent stem cells (iPS), can be used to generate any type of cell found in the body. For us, this means we can use a skin sample from an MND patient to grow motor nerves, glia and muscle cells – separately and together – in a dish. We can then use these cells to model aspects of MND-associated biology and what might go wrong with these cells in patients. We can study how the cells behave in isolation and how they interact with each other. For example, recent work showed that glial cells generated in the lab from a patient with an inherited form of MND were themselves affected by the inherited gene mutation, irrespective of any effect on motor nerves4. Many researchers are now using MND patient-derived stem cells like this to gain insight into disease processes. However, there are still teething issues to overcome. Keeping specialised human cells alive in a dish long enough for them to mature and for us to perform meaningful experiments can be very challenging5. Nonetheless, useful results are already emerging from iPS cell research. These include the discovery of a common genetic abnormality that links some cases of sporadic (non-inherited) MND with many cases of familial (inherited) MND6. This finding has shifted the focus of current laboratory work and already, iPS technology is being used to ask how this genetic abnormality might lead to the death of motor nerves7.
Screen-saver
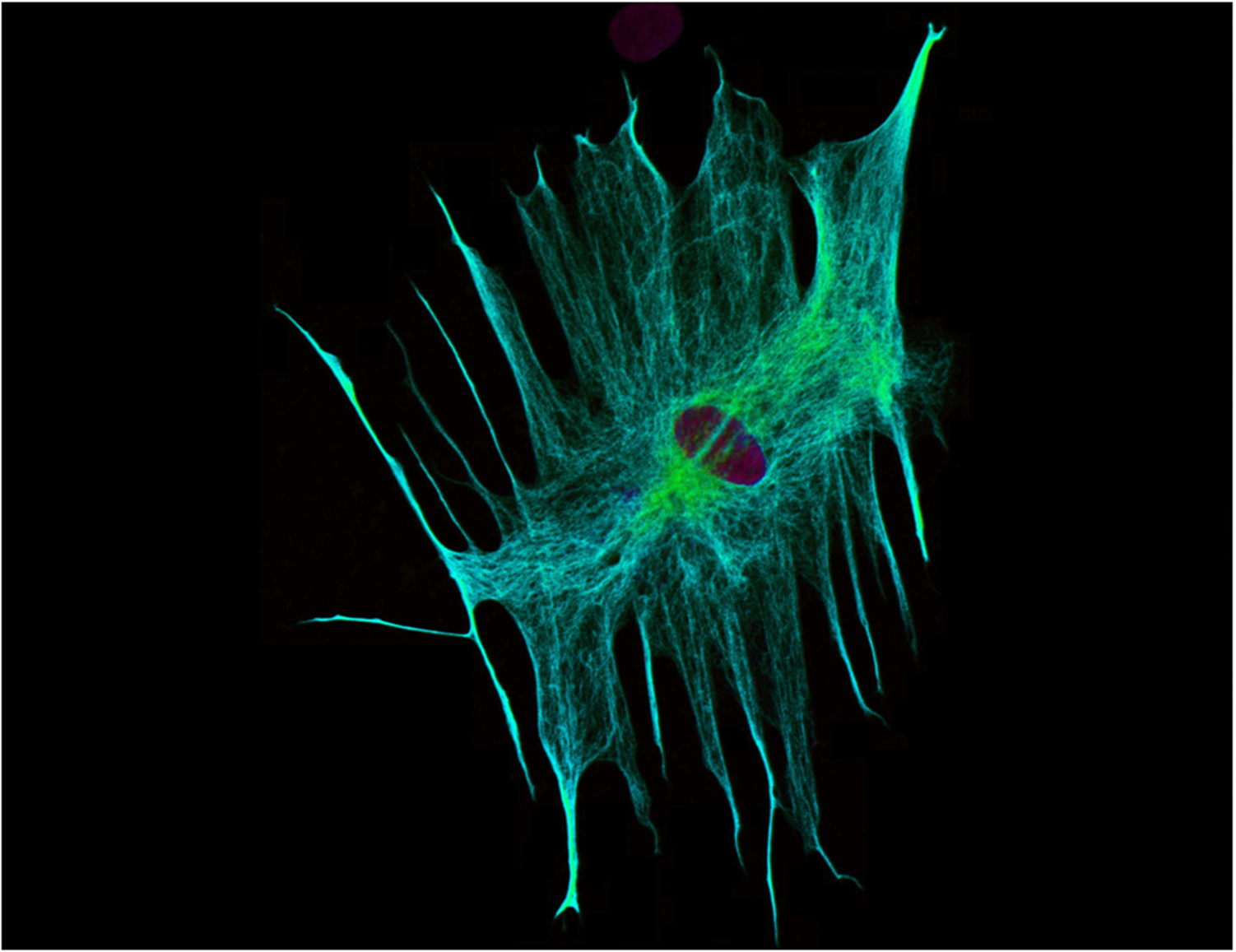
Image: Nina Rzechorzek, MRC Centre for Regenerative Medicine, University of Edinburgh
Drug testing, using human cells in a dish, can now be ‘high throughput’: many thousands of potential drugs can be tested – or ‘screened’ – in the same experiment using the same cells. This means we retrieve an enormous amount of reproducible data describing how each substance affects the cells, much faster than ever before. Combined with iPS technology, we can test chemical compounds directly on patient-derived motor nerves or other relevant types of cells. This doesn’t just tell us what might work as a treatment, it also tells us what is likely to be safe, toxic or have adverse side effects in human cells under certain conditions. This safety aspect is important because a safe compound that fails to treat one particular condition or type of cell might be a useful candidate for treating another human disorder. Overall, this screening process reduces the waste and cost associated with developing new drugs ‘from scratch’. It also provides a human-specific test system that can help minimise the number of animal studies required before clinical trials are carried out in patients. High-throughput drug screening cannot yet replace the need for animal-based experiments, but it catalyses the drug discovery process and in the future it could provide the basis for bespoke patient-targeted therapies.
Replacing the irreplaceable
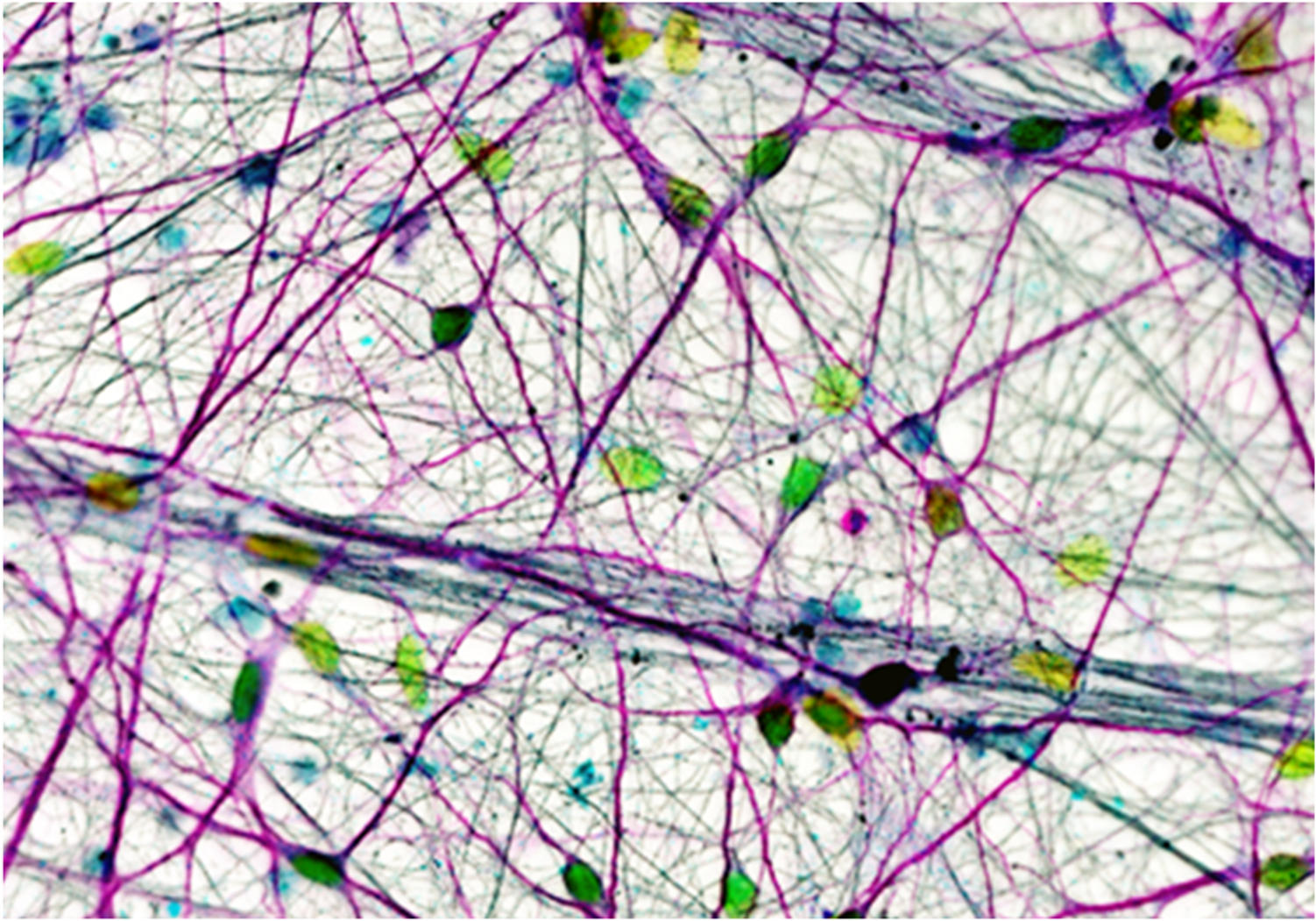
Image: Nina Rzechorzek, MRC Centre for Regenerative Medicine, University of Edinburgh
The idea of replacing lost or damaged motor nerves with new ones by transplanting the patient’s own stem cells is plausible, but very challenging and not yet proven to work. Apart from the complexity of many different cells contributing to MND, any healthy cell placed into the body when it is affected by disease may not be well supported to survive and integrate effectively. Nevertheless, a few studies have shown that such a technique can be safe and potentially beneficial, even though the cells administered to the patient do not necessarily turn into new motor nerves. One theory is that adding certain types of stem cells helps to stimulate ‘endogenous’ repair mechanisms. In essence, the idea is that the transplanted cells ‘kick-start’ the patient’s own nervous system into repair mode, for example by producing supportive cells that release proteins that encourage nerve cell growth8. In this way, the transplanted cells might be used to ‘sculpt’ the environment of motor nerves within the body, making it more favourable for their survival. Many more laboratory-based studies and, eventually, clinical trials are needed before such techniques could become available for MND patients - but they show some promise, especially with regard to slowing the progress of the disease.
Patient power
Never before have patients had such an opportunity to invest in their own future and drive the research to its inevitable success. Euan MacDonald is an extraordinary ambassador for this. Diagnosed with MND at just 29, Euan has worked tirelessly with his family and supporters to keep the research wheels in motion. Since its inception in 2007, The Euan MacDonald Centre for MND Research has provided a ‘hot-bed’ for neuroscientists to explore MND from every angle – including using stem cells to study disease processes in the lab. I am humbled by their efforts – and their determination to get answers for very difficult questions.
The ‘advancing science, building hope’ ethos is central to The Clinic, and to what we strive for on a daily basis. Like iPS reprogramming, scientific advance needs just four key ingredients:
- A team effort – with a team that includes patients, carers, clinicians, scientists, funding bodies, industry, all working towards a common goal
- The right questions – answered robustly, with research of the highest quality
- Hard work – focus, perseverance and creativity in equal measure
- Faith – in the scientific method and the outcomes it will deliver
Whilst the biology may be complex, the concepts are straightforward – understand how motor nerves die and you make a huge leap towards stopping this process and reversing it. But the answers cannot come all at once. Bit by bit, through collaboration and co-ordinated effort the pieces of the puzzle will come together.
Now for the confusing part – I’m a vet. And yes, animals also suffer from motor nerve disorders. And no, we don’t know the exact mechanisms involved, nor do we have any effective treatments to offer our patients. This is the frustration that drove me into research. But iPS technology can offer hope to veterinary and human medicine alike. More than that – stem cell research is bridging the gap between these areas and accelerating discovery on both sides of the species ‘fence’. This will also give us insight into why MND studies on animals have not resulted in effective treatments for human patients. Knowledge transfer between different disciplines and between labs across the world is the fastest route to drug discovery - after all, neurodegenerative disorders are a global problem, requiring a global effort. The parallel explosion in communication and computing technologies means we can realise the full potential of the stem cell revolution – making these truly exciting times.
Useful links
- EuroStemCell fact sheet on stem cell research and MND
- The MND Association (UK) for further information and support
- Euan MacDonald Centre
- Anne Rowling Clinic
- MRC Centre for Regenerative Medicine
Acknowledgements
Image of human iPS cell colony by Karen Burr. Remaining images by Nina Rzechorzek. With thanks to Prof Siddharthan Chandran and Rebecca Devon for their support in preparing this text.
References
- Sydney Brenner – Nobel Laureate in Physiology or Medicine 2002.
- Musaro, A. Understanding ALS: new therapeutic approaches. FEBS J. 280, 4315-4322 (2013).
- Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
- Serio, A. et al. Astrocyte pathology and the absence of non-cell autonomy in an induced pluripotent stem cell model of TDP-43 proteinopathy. Proc. Natl. Acad. Sci. USA 110, 4697-702 (2013).
- Thomson, S. R., Wishart, T.M., Patani, R., Chandran, S. and Gillingwater, T.H. using human induced pluripotent stem cells to model human muscular connectivity: promise or reality? J. Anat. 220, 122-130 (2012).
- DeJesus-Hernandez, M. et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron 72, 245-256 (2011).
- Donnelly, C.J. et al. RNA toxicity from the ALS/FTD C9ORF72 expansion is mitigated by antisense intervention. Neuron 80, 415-428 (2013).
- Gowing, G. & Svendsen, C.N. Stem cell transplantation for motor neuron disease: current approaches and future perspectives. Neurotherapeutics 8, 591-606 (2011).
Last updated: